
Answered Complete the table below for these two… bartleby
Science Chemistry Chemistry questions and answers For the following compound: SeO32- Draw the Lewis Structure Determine the geometry Determine whether the compound is polar or non-polar This problem has been solved! You'll get a detailed solution from a subject matter expert that helps you learn core concepts. See Answer
Is Sulfur Trioxide (SO3) Polar or NonPolar? Lewis Structure (The
Molecules with dipoles (polar molecules) contain polar bonds. lewis dot structure, molecular shape, atom electronegativity. is ClF2 polar or nonpolar. polar. is SeO3 polar or nonpolar. nonpolar. Hydrogen bonding. An unusually large dipole-dipole interaction, H "bonds" to a lone pair (on N, O, or F) only ones it can bond to.
In So3 Polar Or Nonpolar?
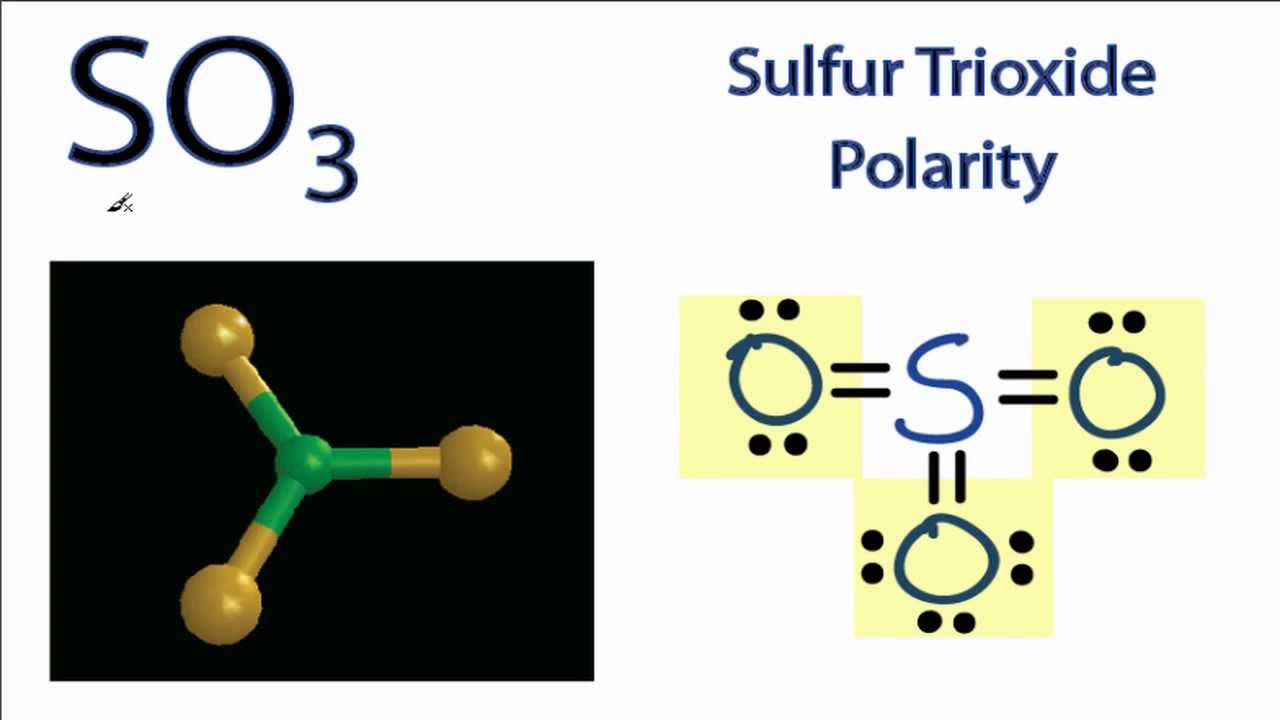
If you look at the Lewis structure for SO3 it appears to be a symmetrical molecule. However, to determine if SO3 is polar we need to look at the molecular.

Best Overview of Polar vs Nonpolar Molecules [No1] Science Education
H2SeO3 ⇌ SeO2 + H2O Let us study its lewis structure, geometry, and hybridization. Contents show Lewis Structure of Selenium Dioxide (SeO2) Also called the Lewis dot structure, it is a pictorial representation of the behavior and arrangement of valence electrons within an atom. It uses dots to represent valence electrons and lines to show bonds.

Determine whether CHCl3 is polar or nonpolar YouTube
SeO3 is a NONPOLAR molecule because all the three bonds (Se=O bonds) are identical and SeO3 has symmetrical geometry which cancels out the bond polarity. Let me explain this in detail with the help of SeO3 lewis structure and its 3D geometry. Why is SeO3 a Nonpolar molecule? (Explained in 3 Steps)
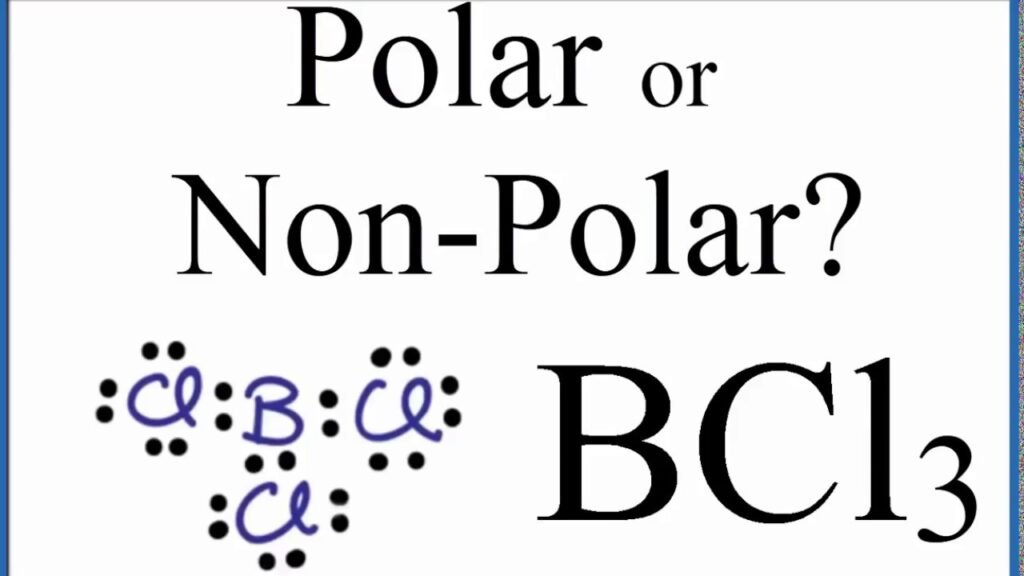
Is BCl3 Polar or Nonpolar?
1 year, 1 month ago Georgia Institute of Technology We don't have your requested question, but here is a suggested video that might help. Classily each molecule as polar or nonpolar: Polur Nonpolar Ansat Bant You must be signed in to discuss. Numerade has step-by-step video solutions, matched directly to more than +2,000 textbooks.

How to Draw the Lewis Dot Structure for SeO3 2 (Selenite ion) YouTube
Also, polar solvents are better at dissolving polar substances, and nonpolar solvents are better at dissolving nonpolar substances. Figure \(\PageIndex{14}\): (a) Molecules are always randomly distributed in the liquid state in the absence of an electric field. (b) When an electric field is applied, polar molecules like HF will align to the.

Is SCN Polar or Nonpolar? Covalent bonding, Molecular geometry
1. Another non polar molecule shown below is boron trifluoride, BF 3. BF 3 is a trigonal planar molecule and all three peripheral atoms are the same. Figure 4.12.1 4.12. 1 Some examples of nonpolar molecules based on molecular geometry (BF 3 and CCl 4 ). Polar molecules are asymmetric, either containing lone pairs of electrons on a central atom.

Figure 2 from Polarnonpolar interconnected elastic networks with
But why? And how can you say that SeO2 is a polar molecule? Want to know the reason? Let's dive into it! SeO2 is a POLAR molecule because the Oxygen (O) present in the molecule is more electronegative, which causes the partial positive (ẟ+) and partial negative (ẟ-) charge to appear on the molecule.

Is CS2 polar or nonpolar? YouTube
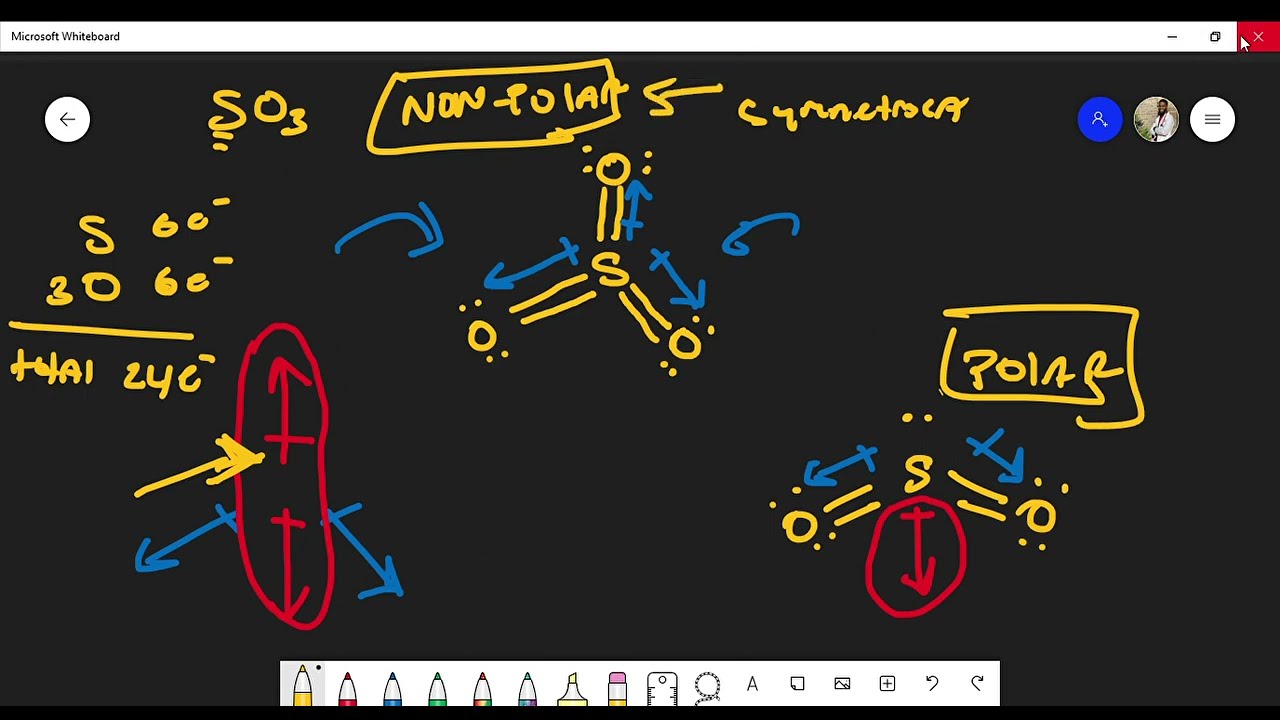
As the bonds (S=O) are symmetrical and the SO3 molecule has a symmetrical geometry, their bond polarity gets canceled with each other. Because of this, there are no positive and negative poles of charges on the overall molecule of SO3. Hence, the SO3 molecule is a nonpolar molecule. I hope you have understood the reason behind the nonpolar.

Polar vs. Nonpolar Bonds — Overview & Examples Expii Ionic Bonding
XeO3 is a polar molecule because it has poles of partial positive charge (ẟ+) and partial negative charge (ẟ-) on it. Let me explain this to you in 3 steps! Step #1: Draw the lewis structure Here is a skeleton of XeO3 lewis structure and it contains three Xe=O bonds.

CH2Cl2 polar or nonpolar Science education, Learning science, Science
It also discusses if SeO3 is polar or nonpolar in addition to the bond angle, hybridization, and the molecular geometry. This video shows you how to draw the lewis dot structure of SeO3 also known as selenium trioxide. It also discusses if SeO3 is polar or nonpolar in addition to the bond angle, hybridization, and the molecular geometry..

Is SO3 Polar or Nonpolar? Polarity of SO3
Is SeO3 polar or nonpolar ? Question = Is SeO3 polar or nonpolar ? Answer = SeO3 ( Selenium trioxide ) is Nonpolar What is polar and non-polar? Polar "In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole or multipole moment.

Ch4 Polar Or Nonpolar / Solution Is The Ch4 A Polar Or Non Polar Chemistry
SO3 is nonpolar and this is because of the trigonal planar shape of sulfur trioxide. Sulfur and oxygen have a difference in electronegativity due to which polarity arises in the S-O bond but the three S-O bonds lie at 120 degrees angle with each other cancels out the overall polarity and resulting in the formation of SO3 as a nonpolar molecule.
MakeTheBrainHappy Is CO Polar or Nonpolar?
Want to know the reason? Let's dive into it! SO3 2- is a POLAR ion because the Oxygen (O) atom is more electronegative and it also has lone pair, which results in an asymmetric shape of the ion. Because of this, the partial positive (ẟ+) and partial negative (ẟ-) charge appears on the ion.

Best Overview Is CH3F Polar or Nonpolar Science Education and Tutorials
Molecular Polarity. To determine if a molecule is polar or nonpolar, it is frequently useful to look at Lewis structures. Nonpolar compounds will be symmetric, meaning all of the sides around the central atom are identical - bonded to the same element with no unshared pairs of electrons.Polar molecules are asymmetric, either containing lone pairs of electrons on a central atom or having atoms.